|
12/30/2023 0 Comments Vaccine production companies Lipids suppliers to BioNTech include Merck and Evonik Industries, according to the Wall Street Journal, while Dermapharm in Germany, Acuitas Therapeutics in Canada and Polymun Scientific Immunbiologische Forschung in Austria are involved in the formulation process. ‘The entire supply chain of components and materials that go into these vaccines is new,’ adds Kis, who notes that the supply of some specialised lipids is likely squeezed by intellectual property issues. ‘The manufacturing step is very unusual and tricky,’ Lowe warns. Other drug companies cannot easily chip in. Reports reveal ‘very exotic mixing technology’ to control the shape and composition of the lipid nanoparticles and mRNA, says Lowe. The two mRNA vaccines on the market use commercially available lipids but also ‘proprietary, bespoke lipids’, notes Lowe. This view is shared by Derek Lowe, a drug discovery chemist in the US, who has written about mRNA manufacture in his blog, In the pipeline. ‘Here is the bottleneck, the rate limiting step in the process,’ says Kis. Microfluidics must mix the mRNA and lipids in a controlled environment. Next the mRNA is encapsulated into lipid spheres. ‘The goal is to purify out the RNA to the highest possible degree, because you don’t want any impurities that could lead to side effects,’ explains Kis. This is separated out according to electrical charge or size, using tangential flow filtration or various chromatography methods. The mRNA synthesis takes two hours, while making the vaccine takes a couple of days.Įnzymes are added to degrade the DNA template, leaving behind an RNA molecule between 40 nucleotides long. ‘The reaction mix has a well-defined composition and relatively few components, with no living cells or cell excretions or cell debris,’ says Zoltán Kis, a chemical engineer at the Future Vaccine Manufacturing Hub at Imperial College London in the UK. Making the mRNA itself – which codes for the virus spike protein inside our cells – is uncomplicated. The entire supply chain of components and materials that go into these vaccines is new The nucleotides are made chemically,’ explains immunologist Drew Weissman at the University of Pennsylvania, who contributed to the technology licensed by Moderna and BioNTech. ‘All of the enzymes are made in bacteria. Moderna and BioNTech are reticent on manufacturing specifics, but in general a polymerase follows a DNA template to make the mRNA sequence by linking nucleotides together. Large-scale production of such a vaccine has never happened before. The Vaccines Manufacturing and Innovation Centre will be the UK’s first national vaccines manufacturing and innovation facility mRNA bottlenecksīioNTech–Pfizer’s vaccine is the first mRNA vaccine ever to be approved for people by the FDA. Source: © Richard Pohle/WPA Pool/Getty Images 
Vaccine developers are also adapting existing facilities and partnering with sometime rivals to take advantage of their manufacturing capacity and expertise. Last weekend the British government announced an additional £47.6 million to get the UK Vaccine Manufacturing and Innovation Centre being built at the Harwell Science and Innovation Campus, online even sooner. Building additional capacity is one way companies and governments are approaching the challenge. And as Matt Hancock, the UK’s health secretary, told the House of Commons on 18 March ‘the process of manufacturing vaccines is complicated, and subject to unpredictability’.Įach vaccine faces production issues the newer technologies are effectively starting from scratch. But those companies that have surmounted the challenges of development now face the next phase: manufacturing doses on an enormous scale. Hot on their heels are rivals based on adenovirus vectors from AstraZeneca and Johnson & Johnson, as well as Sputnik V from Russia.Įarly successes in developing vaccines by upstarts like Moderna and BioNTech papered over the struggles of vaccine heavyweights like Merck, GSK and Sanofi. 
By December 2020, these two had become the first vaccines to be approved by the US Food and Drug Administration (FDA). 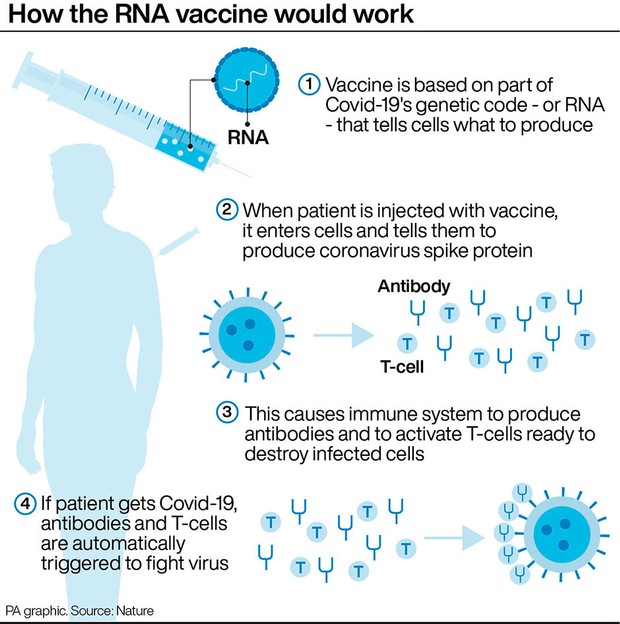
The mRNA candidate from BioNTech and Pfizer followed in April. The first Covid-19 vaccine candidate went into the arms of volunteers in Seattle in March 2020. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
